ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Nida Jamil Khan
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Breast cancer cell lines have been used widely to investigate breast cancer pathobiology and new therapies. Breast cancer is a molecularly heterogeneous disease, and it is important to understand how well and which cell lines best model that diversity.Human breast tumor cell lines are needed for multidisciplinary research in breast cancer.In the laboratory breast cancer is often modelled using established cell lines. Utilization of cell lines as subtype systems in threedimensional and heterotypic cultures represent powerful approaches to investigate the signaling interactions that contribute to breast cancer biology. In the present review we discuss the use of breast cancer cell lines as experimental invitro models and put forward suggestions for improving their utility in translational breast cancer research
Keywords |
| Cell culture, invitro models, primary breast tumors. |
I . INTRODUCTION |
| Cell lines are widely used as invitro models in cancer research.They have numerous advantages as they are easy to handle, exhibit a relatively high degree of homogeneity, can be easily replaced from frozen stocks if lost through contamination and represent an unlimited self replicating source that can be grown in almost infinite quantities.However, cell lines are prone to genotypic and phenotypic drift during their continual culture and this is particularly common in more frequently used cell lines especially the ones that have been deposited in cell banks for many years[1]. |
| The first human cell line has been established in a Baltimore laboratory over 50 years ago by George Gey[2]. This cell line was called HeLa- after Henrietta Lacks, the lady who had cervical cancer and from whom the cell line was derived[3]. The first breast cancer cell line to be established was BT-20 in 1958[4]. However, the most commonly used breast cancer cell line in the world is MCF-7 established in 1973 at the Michigan Cancer Foundation. This cell line is an ideal model to study hormone response due to its exquisite hormone sensitivity through expression of oestrogen receptor[5]. |
II. PRIMARY CULTURE OF BREAST TISSUE |
| Before the cell lines were established primary cultures of cells obtained directly from breast tumors were prepared.A detailed pathology of these cultures was available to allow the characteristics of the culture to be compared to original tumor.However they had their own set of limitations. They had slow population doubling time and a finite lifespan before senescence.These limitations paved way for the use of cell lines in translational breast cancer research. |
III. BREAST CANCER CELL LINES |
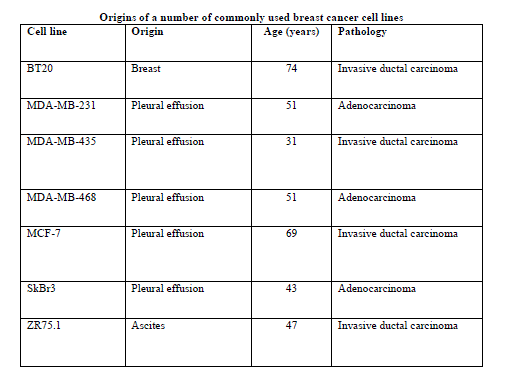
| Origins of some commonly used breast cancer cell lines are listed in table 1.The table clearly shows that these long established cell lines are not derived from primary breast tumors, but from tumor metastasis , especially aspirates or pleural effusions. Thus, research that relies on such cell lines will be biased towards more rapidly progressive type of breast carcinoma and to late stage disease, rather than lower grade and earlier stage breast cancers. However, there is evidence that gene-expression profiles of primary tumours and their metastatic counterparts are not markedly different [6-8]. |
 |
| Apart from these traditional cell lines,a number of novel breast cancer cell lines have been established and characterized in recent years[9-12]. Some of these have the advantage of being established from a primary lesion rather than from a distant metastasis.Clearly these new cell lines are attractive models, and some of these have been deposited in recognized cell banks (e.g. American Type Culture Collection ; http://www.atcc.org) and are now commonly available to breast cancer researchers. |
V. THE CELL CULTURE ENVIRONMENT |
| 2D VS 3D CULTURE |
| Complex inter-relationships that exist between cells in vivo are lost when cell lines are cultured on plastic in two dimensions. In addition, cell lines are often sensitive to culture conditions – particularly the inclusion of growth factors that can sometimes alter the cell phenotype, resulting in inappropriate pathway activation or differentiation. Culture under inappropriate conditions can also dramatically influence cell morphology, cell–cell and cell–matrix interactions, cell polarity and differentiation[13,14], as well as altering signalling cascades and gene expression[15]. Identification of the most appropriate conditions to maintain the desired cell phenotype is thus a critical issue. As well as considering the molecular profiles of breast cancer cell lines, we also need to look beyond simple two-dimensional breast cancer models. There has thus been a shift in growing cells in more physiologically relevant three-dimensional systems with the increased complexity of including multiple cell types [13,16]. |
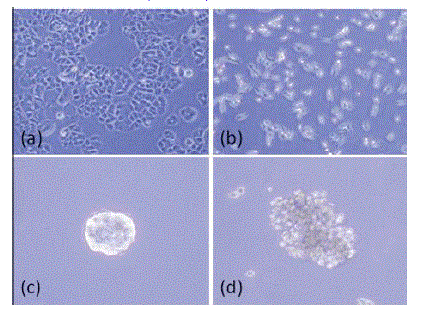
| As highlighted by Kenny and colleagues, cell morphology in three dimensions is different from that observed in two dimensions on tissue culture plastic[17]( fig 1). In two dimensions, luminal-like epithelial cells demonstrated the classic cobblestone morphology and expression of cell–cell adhesion molecules such as E-cadherin, whereas basal epithelial cells displayed a more elongated and spiky appearance and expressed markers of epithelial–mesenchymal transition such as vimentin. In contrast, cell lines grown in three-dimensional culture showed four different morphologies: round, mass, grape-like and stellate [17]. |
| Three dimensional models have also provided an insight into the biology of breast cancer by implicating a role for β1 - integrin in breast cancer progression and by use of blocking antibodies to reverse the malignant phenotype of epithelial cells[18]. With the role of the stroma in regulating breast cancer behaviour receiving increased attention [19-21] and the recent recognition that basal and luminal breast cancers behave very differently when co-cultured with stromal fibroblasts[22], other threedimensional breast cancer models have incorporated stromal cells such as fibroblasts[23], macrophages[24] and endothelial cells [25]. |
| These complex multicellular three-dimensional cultures are not just a tool for understanding disease progression, but may have important implications in drug screening. This was highlighted recently by Pickl and Ries[26], who demonstrated a significantly higher response of SKBR3 cells to trastuzumab when the cells were cultured in three dimensions compared with cells cultured in two dimensions. Three-dimensional models may thus become a more widespread tool for research and drug screening, and while these models are technically challenging to establish, they are in the long term much more biologically relevant models for studying the disease in vitro. |
 |
HOMOTYPIC VS HETEROTYPIC CULTURES |
| Another constraint of cell culture experiments is that they are usually homotypic and do not allow the examination of tumour–stromal cell interactions. During tumour growth the stroma evolves to establish paracrine interactions that facilitate tumour progression. Breast cancer cell lines have been used to build heterotypic co-culture systems that allow the selective examination of interactions between tumour and stromal cells, including macrophages, fibroblasts and endothelial cells. For example, co-cultures have been used to investigate the role of macrophages in promoting breast tumour cell invasion, and these experiments have allowed the delineation of the signalling pathways that mediate crosstalk between breast tumour cells and macrophages[26-28]. Therefore, the use of co-culture systems may provide new stromal targets to inhibit tumour progression[29]. |
V. SCOPE OF DEVELOPMENT IN BREAST CANCER CELL LINES |
| Although there are now a reasonable number of breast cancer cell lines available to reflect the molecular sub-groups, relevant models are lacking for some of the rarer histopathological types.Further there is no known cell line derived from male breast cancer and, given that the incidence of male breast cancer is rising [30], this poses a challenge for modelling in a functional sense. |
VI. CONCLUSION |
| Although more technically demanding to establish, if it is properly characterized, primary culture offers a more relevant clinical model of this disease that is likely to provide more meaningful data. It also offers a better route to cytogenetic analysis. But because of their ease of use, there is no doubt that established cell lines will continue to be used as models for breast cancer. Some of the more recently established novel cell lines, particularly those established from primary breast tumours and now available through conventional cell line repositories, are worth considering as research tools. Tremendous advances in our understanding of the biology of breast cancer have been made over the past several decades using breast cancer cell lines. We must now move beyond the „one marker, one cell lineâÃâ¬ÃŸ studies of the past and use knowledge gained through genetic and transcriptomic profiling to use cell lines or cell line panels more effectively as experimental models to study specific subgroups of breast cancer, because this is likely to have the greatest impact on improving outcome for breast cancer patients. |
References |
|