ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
|
Dr.K.Suneetha Faculty, Department of Biochemistry, Acharya Nagarjuna University, Guntur, Andhra Pradesh, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
The freshwater fish Labeo rohita was exposed to endosulfan and fenvalerate a synthetic pyrethroid. The LC50 values determined for endosulfan and fenvalerate at 24hrs were 0.6876 and 0.4749 μgl-1 respectively. The fish were exposed to lethal endosulfan 0.6876 and fenvalerate 0.4749 μgl-1 and sublethal concentrations of endosulfan 0.06876 and 0.04749 μgl-1 for 24hrs and 15days. Changes in the Total proteins and protein metabolic enzymes such as AAT and ALAT of vital organs such as brain, liver, gill, kidney and muscle were studied. The percent decrease of total proteins and percent increase in activities of AAT and ALAT were observed. more pronounced in the fenvalerate exposure than endosulfan
Keywords |
| Endosulfan, Fenvalerate, Total proteins, AAT and ALAT. |
INTRODUCTION |
| The pesticides have been recognized as one of the serious pollutants of the aquatic ecosystems with deleterious effects on the living resources. Increasing number and amount of industrial, agricultural and commercial chemicals discharged into the aquatic environment having led to various deleterious effects on the aquatic organisms [4]. Aquatic organisms, including fish, accumulate pollutants directly from contaminated water and indirectly via the food chain [17]. Understanding of the protein components of cell becomes necessary in the light of the radical changes that take place in protein profiles during pesticide intoxication. Both the protein degradation and synthesis are sensitive over a wide range of conditions and show changes to a variety of physical and chemical modulators. The physiological and biochemical alterations observed in an animal under any physiological stress can be correlated with the structural and functional changes of cellular proteins. Proteins occupy a unique position in the metabolism of cell because of the proteinaceous nature of all the enzymes which mediate at various metabolic pathways [7, 6]. Pesticides also influence the activity of different enzymes. A slight variation in enzyme activities would affect the organism [13]. Aminotransferases mobilize the aminoacids into carbohydrate and lipid metabolism. There exists a rapid turnover of free aminoacids from cell to cell, tissue to tissue through the circulating fluid and utilize for various purposes through interconversions [20]. Transminases form an important group of enzymes mediating carbohydrates, protein and lipid metabolism. Transmination represents the mechanism causing eventual deposition of nitrogenous waste products like ammonia and urea resulting in the production of carbon compounds, which contribute towards gluconeogenesis and fattyacid formation. Therefore in the present study, an attempt has been made to explore the effects of endosulfan and fenvalerate on the protein metabolic alterations in different tissues of fish Labeo rohita. |
II.MATERIALS AND METHODS |
| The freshwater fish Labeo rohita (Hamilton) of size 6-7 ±1cm and 6-8 g weight were brought from a local fish farm and acclimatized at 28 ± 2 0C in the laboratory for one week. The stock solutions for Endosulfan 35% Emulsifiable Concentrate (EC) and fenvalerate 20% Emulsifiable Concentrate (EC) were prepared in 95% acetone to yield a concentration of 100mg100ml-1 which was further diluted with distilled water to get a working solution. The water used for acclimatization and conducting experiments was clear unchlorinated ground water. In each test ten fish were introduced in toxicant glass chambers with a capacity of ten liters. The data on the mortality rate of fish was recorded. The dead fish were removed immediately. The toxic tests were conducted to choose the mortality range from ten percent to ninety percent for 24hrs in static tests. The concentration that produced fifty percent mortality in test species noted. LC50 values were calculated by Finney’s Probit analysis [3]. Total protein content was determined with Folin-Ciocalteau reagent according to Lowry method[8]. Assay of aspartate aminotransferase and alanine aminotransferase: The activity of Aspartate aminotransferase and Alanine aminotransferase was determined by the method of Reitman and Frankel [12]. Aspartate aminotransferase 0.5 ml of buffered substrate (Aspartate, - ketoglutaric acid) was added to 0.1 ml of crude enzyme extract and placed in water bath at 37ºC. To the blank tubes, 0.1 ml distilled water was added instead of extract. After 1 h, 2 drops of aniline citrate reagent and 0.5 ml of 2-4 Dinitrophenylhydrazine (DNPH) reagents was added and kept at room temperature for 20 min. Finally, 5.0 ml of 0.4N sodium hydroxide was added. A set of standards also treated in the same manner and read at 520 nm after 10 min. The results were expressed in μmoles of pyruvate formed min-1 mg protein-1 h-1. Alanine aminotransferase 0.5 ml of buffered substrate (Alanine, - ketoglutaric acid) was added to 0.1 ml of crude enzyme extract and placed in water bath at 37ºC. To the blank tubes, 0.1 ml distilled water was added instead of extract. After 1 h, 2 drops of aniline citrate reagent and 0.5 ml of 2-4 Dinitrophenylhydrazine (DNPH) reagent was added and kept at room temperature for 20 min. Finally, 5.0 ml of 0.4 N sodium hydroxide was added. A set of standards also treated in the same manner and read at 520 nm after 10 min. The results were expressed in μmoles of pyruvate formed min-1 mg protein-1 h-1. |
III. RESULTS AND DISCUSSION |
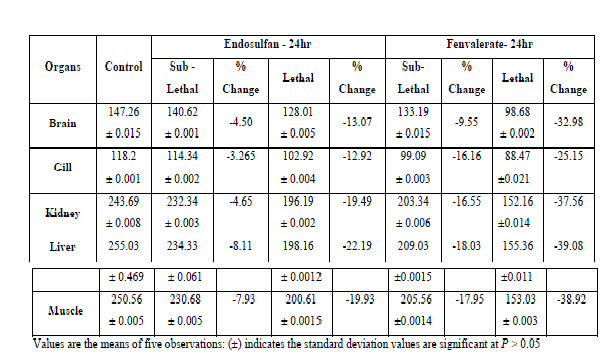 |
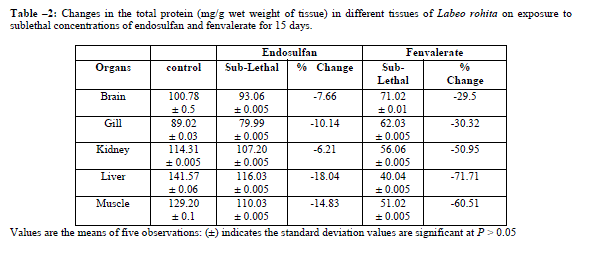 |
| Under lethal and sublethal exposure to endosulfan and fenvalerate the total protein content was found to decrease in all the tissues of test fish compared to control (Table-1,2). Since fish have a very little amount of carbohydrates so next alternative source of energy is protein to meet the increased energy demand [14]. Reference [2] stated that the concomitant reduction in proteins supports the view that a pesticide exerts a proteolytic effect. Reference [21] observed the total protein depletion in tissues of muscle, liver, kidney, brain and gill of fish Labeo rohita exposed to both technical as well as 20% EC of chlorpyrifos. Reference [23] observed the decrease in protein content in the tissues of brain, liver and muscle of fish Clarias batrachus in sublethal concentration of endosulfan and fenvalerate. Reference [5] observed the depletion of proteins under exposure to kelthane an organochlorine insecticide in the freshwater fish Channa punctatus. Reference [19] observed significant decrease in the protein content under sublethal concentrations of alachlor in Channa punctatus. Decrease in total proteins in liver, muscle, kidney and gills under dimethoate toxicity in fish Arias Dussumieri was observed [11]. |
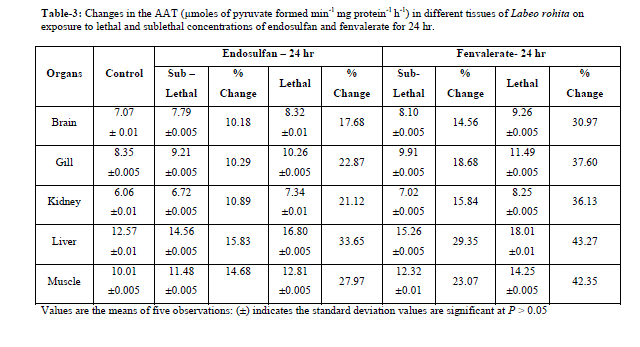 |
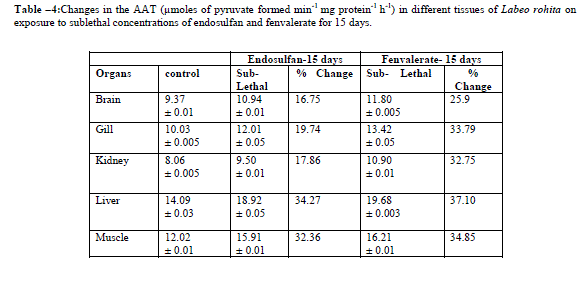 |
 |
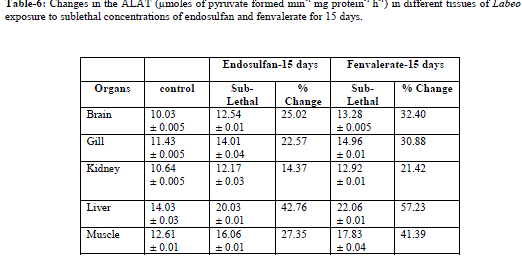 |
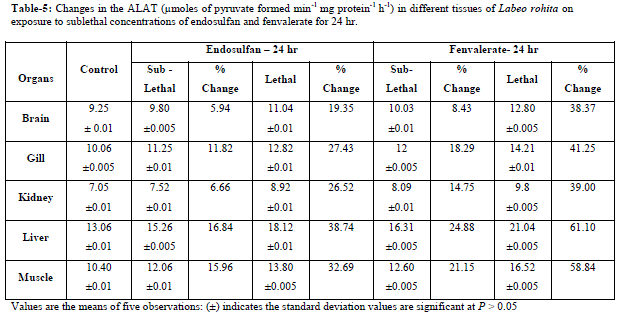
| Values are the means of five observations: (±) indicates the standard deviation values are significant at P > 0.05 Increased activities of AAT and ALAT was observed in both lethal and sublethal concentrations of endosulfan and fenvalerate for 24hrs and 15 days (Tables-3,6). Elevation of AAT and ALAT in different tissues of fish suggests either increased operation of transamination or increased synthesis of amino acids from other sources like glucose or fattyacids during endosulfan and fenvalerate intoxication. In the present study, ALAT activity is found to be relatively higher than AAT in all tissues of control and test fish, suggesting pyruvate contribution is slightly more than oxaloacetate formation [14]. The increase in activities of aminotransferases as observed in the present study were in agreement with earlier reports, demonstrating a consistent increase in the activities of these enzymes under conditions of enhanced gluconeogenesis. Reference [10] observed increase in levels of SGOT and SGPT activities under sublethal effect of Dichlorvos in Channa gachua. The activities of serum GOT and GPT increased in Oreochromis niloticus following lead pollution [9]. The same trend was observed in liver and muscle of Colisa fasciatus due to toxicity of ethanolic extract of Nerium indicum mill (Lal kaner)latex by [22]. Increased in activities of AAT and ALAT were observed in Channa punctatus under alachlor exposure [19]. Reference [16] reported induction in AAT and ALAT in liver and blood of Labeo rohita after exposure to endosulfan. |
VI. CONCLUSION |
| The depletion of proteins under the stress of endosulfan and fenvalerate toxicity observed in different tissues of Labeo rohita indicates the proteolysis, prompting the suggestion that the proteins were utilized to meet the excess energy demands imposed by the toxic stress. An increase in the activities of AAT and ALAT was recorded indicating that there was an increased demand for energy due to tissue impairment. Contrariwise, elevation of AAT and ALAT reflect hepatic disease, some inflammatory disease or injury to the liver- hepatocellular damage [1 and 18]. The increase in the activities of AAT and ALAT as observed in the present study may also be due to the mitochondrial disruption and tissue damage as a result of endosulfan and fenvalerate induced stress. |
References |
|