e-ISSN:2320-1215 p-ISSN: 2322-0112
e-ISSN:2320-1215 p-ISSN: 2322-0112
1Dr. B.C. Roy College of Pharmacy & Allied Health Sciences (BCRCPAHS), Durgapur - 713206, West Bengal, India.
2Chemical Technology Dept., Calcutta University, Kolkata-700 009, West Bengal, India.
3Fresenius Kabi Oncology Ltd., Kalyani,-741235, West Bengal, India.
Received: 02/06/2013; Revised: 24/06/2013; Accepted: 02/07/2013
Visit for more related articles at Research & Reviews in Pharmacy and Pharmaceutical Sciences
5-Fluorouracil (5-FU) is widely used as an anti-cancer drug, but causes severe side effects. Controlled release systems could be useful to keep the concentration of 5-FU at a low level, so that the side effects can be reduced. The purpose of this research study was to formulate transdermal film loaded with 5-Fluorouracil by solution casting method using ethyl cellulose, HPMC E15 and HPMC K4M in various combinations and to investigate the effect of different polymer on drug permeation and other physicochemical characteristics of the film. The in-vitro study using dialysis membrane was carried out and revealed that HPMC E15 can exhibit more sustained formulation than HPMC K4M. Further, pharmacological evaluation of the film was also investigated on EAC induced tumors in Swiss albino mice. The above in-vivo investigation showed highly significant inhibition of tumor volume, packed cell volume and viable tumor cell count with the simultaneous increase in percentage of non viable cell count and increase in life span in EAC bearing mice. The results thus revealed a positive response and support the promising antitumor effect of formulated 5-FU film.
transdermal film, controlled release, in-vitro & EAC.
Transdermal drug delivery system is the modern delivery system to deliver the drug by by-passing the first-pass metabolism problem. It is used to deliver the drug through the skin to systemic circulation. Currently more than 35 TDDS products are approved in United States for the wide variety of condition like hypertension, angina, motion sickness, severe pain, local pain etc.
The non- invasive character of TDDS makes it accessible to a wide range of patient population and a highly acceptable option for drug dosing. 5-FU is an antimetabolite with promising antineoplastic activity against several premalignant & malignant condition of the skin. It is also shown to be active against a variety of solid tumors including those in breast, colon, rectum and cervix [1,2]. Transdermal delivery of 5-FU may overcome certain limitations associated with oral and parenteral administration. Its oral administration shows significant variation in oral bioavailability, ranging between 0-80% [3] and in case of parenteral administration the main problem is rapid elimination of the drug with apparent half-life of 8-20 min for that reason we have targeted to formulate transdermal film using 5-Fluorouracil as the model drug.
Here, we have attempted to optimize the suitable ratio of the hydrophobic and hydrophilic polymer to formulate the film by studying the permeation of the drug through dialysis membrane (in-vitro).
5-Fluorouracil was purchased from Sigma Aldrich. Ethyl cellulose, HMPC E15, HMPC K4M, oleic acid and glycerin were also purchased from Loba Chemie Pvt. Ltd,Mumbai. Dialysis membrane from HIMEDIA Lab. All other solvents and chemicals used were of analytical grade.
Experimental tumors have great importance in modeling and Ehrlich ascites carcinoma (EAC) is one of the commonest tumors. EAC is referred to as an undifferentiated carcinoma and is originally hyper diploid, has high transplantable capability, no-regression, rapid proliferation, shorter life span, 100% malignancy and also does not have tumor-specific transplantation antigen (TSTA).
The in- vivo study was performed as per the guidelines approved by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Social Justice and Empowerment, Government of India. The Institutional Animals Ethical Committee of BCRCPAHS, (BCRCP/IAEC/4/2012) approved the protocol for the present study.
Four groups of Swiss Albino Mice, each containing six healthy mice of the same sex (female in this case), approximately of same age ( 10 weeks) and average body weight (18-20 g), were selected at random and housed in poly acrylic cages with not more than six animals per cage and maintained under laboratory conditions(25-27°C). One of these four groups served as control while the others are normal and as test. They were allowed free access to standard dry pellet diet and water at libitum. The animals were acclimatized to laboratory conditions for one week before commencement of the experiment.
Transdermal film of 5-FU was prepared by solvent casting method. Here the polymeric solution was prepared by dissolving EC, HPMC K4M, and HPMC E15 in 20 ml mixture of acetone and chloroform (3:1) stirred on magnetic stirrer for 10 min for complete dispersion of the polymers. Then 20 mg of 5-FU was added to it and stirred again for 10 min for complete dispersion. Then 10% glycerine and 5% oleic acid were added to it. After stirring for 5 min the solution was poured on a 6.06 cm diameter Petridis. Then the solvent was allowed to evaporate under ambient temperature (32°C, 45%RH) for 24 hr. The prepared film was then scraped for evaluation.
First a polymer combination was selected to produce the film without the drug. The combination of the formulations is shown in table1. We have denoted the formulations with PF.
From pre formulation study, following eight formulations were selected as the final formulations.
Before going to the final formulations first IR study of the physical mixture of the drug, EC, HPMC K4M and HPMC E15 was performed to investigate the compatibility.
After the pre formulation study and after conforming that there is no drug-excipient interaction, the following formulations were prepared (table 2).
The prepared films were evaluated for their physical appearance, uniformity of thickness, weight variation, folding endurance, drug content, moisture content, moisture uptake, flatness and in- vitro release study across the dialysis membrane.
Weight variation was determined by individually weighing 6 randomly selected films and calculating the average weight and standard deviation. The individual weight should not deviate significantly from the average weight [4].
Screw gauge was used to determine thickness of the films. It was placed at three different positions by keeping the film in between two glass slides of known thickness and average thickness was calculated and the values are given in table-4.
The folding endurance was measured manually. A strip of film having an area of 2cm2 was cut evenly and repeatedly folded at the same place till it broke/cracked. The number of times the film could be folded at the same place without breaking/cracking gave the exact value of folding endurance and the results are reported in table-4 [5,7].
The prepared film were weighed individually and kept in a desiccator containing silica gel at room temperature for 24 h. The films were weighed again after a specified interval until they show a constant weight.The percent moisture content was calculated using following formula [6] .
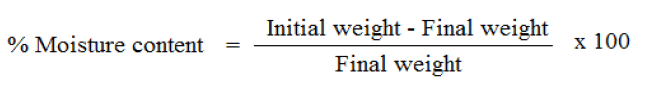
Weighed films were kept in a desiccator at room temperature for 24 h. These were then taken out and exposed to75.3% relative humidity using saturated solution of sodium chloride in a desiccator until a constant weight is achieved. Percent moisture uptake is calculated as given below [6].
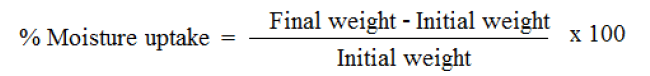
A transdermal film should possess a smooth surface and should not constrict with time. This can be demonstrated with flatness study. For flatness determination, one strip is cut from the centre and two from each side of film. The length of each strip is measured and variation in length is measured by determining percent constriction. Zero percent constriction is equivalent to 100 percent flatness.
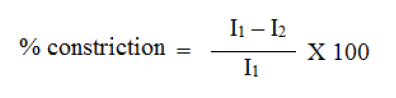
I2 = Final length of each strip
I1 = Initial length of each strip
The in-vitro release study was done by the Keshary Chien diffusion Cell method. In this method transdermal film was placed in between receptor and donor compartment of the diffusion cell. The transdermal system faces the receptor compartment in which receptor fluid i.e., buffer was placed. The whole assembly was kept on magnetic stirrer and solution in the receiver compartment was constantly and continuously stirred with 600 rpm throughout the experiment using magnetic beads. The temperature of receptor compartment was maintained 37 ± 2°C. At predetermined time intervals, the 5ml receptor fluid was removed for analysis and was replaced with an equal volume of phosphate buffer pH 7.4. The concentration of drug was determined spectrophotometrically at 266 nm wavelength with suitable dilution [8,9] .
In-vitro permeation studies were carried out using modified Keshary Chien diffusion cell. The dialysis membrane was previously soaked for 24 hours in distilled water. The film were adhered to the barrier membrane (dialysis membrane, which was dipped in phosphate buffer, pH7.4for over night) and the membrane was tied firmly to the donor compartment of the Keshary Chien diffusion cell, the receptor compartment of which was filled with 57 ml phosphate buffer. The donor compartment was lowered to the receptor compartment in such a way that the dialysis membrane just touches the media of the receptor compartment. The total setup was placed on a magnetic stirrer. The temperature of receptor compartment was maintained 37 ± 2°C. The content of the diffusion cell was stirred using a Teflon coated bead at a constant speed (600 rpm). Samples were withdrawn (5 ml) at predetermined time intervals and replaced with same amount of phosphate buffer to maintain the sink condition. After suitable dilution, the samples were analyzed for drug content using UV-VIS 1800 spectrophotometer (SHIMADZU) at wavelength of 266 nm. The permeation study was carried out for 7 hours [10] .
The stability study of the formulations was done according to ICH guide line Q1A specifications in stability chamber. We performed the stability study in intermediate & accelerated conditions in closed containers (Table 3). In both cases sampling were done 3 times i.e., 0, 3 & 6 months and performed test for the drug content uniformity.
The transdermal drug delivery system of 5-FU was prepared using the polymers EC, HPMC E15 and, HPMC K4M, using glycerin as a plasticizer by solvent casting technique. The prepared films were evaluated for physicochemical parameters with in-vitro drug permeation studies. The selection of polymer combinations produces clear, smooth, uniform, substantive, flexible and desired thickness film for the transdermal drug delivery systems of 5-FU.
Characteristics of the formulation were studied in in-vitro conditions. The permeation study was carried out in phosphate buffer (pH 7.4) for 7 hours, in order to find out the diffusion mechanism which predominately influences the drug permeation from the membrane. The drug content uniformity and the mass uniformity of the prepared formulations have shown that the process used to prepare the films in this study was capable of giving films with uniform drug content and the thickness of the films varied from 0.0855±0.005 to 0.134±0.003 mm.
The percentage moisture uptake and percentage moisture content of the formulations increases with addition of the HPMC with EC. The formulation FF2 with 1:1 EC & HPMC K4M showed highest % of moisture content and the moisture content decreased with increase in the concentration of HPMC E15. This proved that the moisture content increased due to the admixture of HPMC. Between the two grades of HPMC we observed that HPMC K4M showed more hydrophilic nature than HPMC E15.
The folding endurance of the films varied from 72±1.67 to 144.5± 3.2 folds. From the results obtained it was found that the folding endurance decreased with the addition of the HPMC. Again, increased concentration of the HPMC K4M provided higher folding endurance than HPMC E15 with equal amount of EC, this may be due to the higher moisture holding capacity of the polymer K4M.
Now, to understand the release profile, first we made in-vitro release study using dialysis membrane. From the Cumulative % release in 7 hr (Table 5), we observed that the formulation FF1 showed very slow release of 56.76% in 7hr. The FF2 showed highest % release of 96.53 %. The FF1 showed lowest % release may be due to the release retarding effect of the EC and FF2 showed 96.53 % release due to the combination of HPMC K4M & EC. Again, if we study the formulation FF3 which is 1:1 combination of EC & HPMC E15 we observe that it released the formulation slower than FF2 but higher than the rate of FF1and for the formulations of FF4 to FF8 we prepared the formulations in combination of three polymer EC, HPMC K4M with HPMC E15 and we observed as the concentration of HPMC E15 increased with a decrease in concentration of HPMC K4M the % release of the drug through dialysis membrane decreased accordingly. That means in between two grades of HPMC, E15 provides more sustained release than K4M.
To understand the nature of release we fitted the drug release data to Zero order, First order & Higuchi model and tabulated the R2 Value in each case.
We observed that all formulation showed highest linearity to the Higuchi model.
From the cumulative % release data we discarded FF1, FF2 and FF4 for further study. FF1 was discarded because it provided very slow release. FF2 & FF4 , the release rate was faster which is undesirable in case of a transdermal film formulation.
To understand the release mechanism we fitted the R2 values to the Korsmeyer-Peppas Model to find out the ‘n’ values.
Then we observed that all formulations showed non-fickian release mechanism. Therefore, the release of drug 5-FU from the film takes place by diffusion and polymeric chain erosion.
The method followed here had long been standardized in our associated laboratory at Jadavpur University, Kolkata [11,12,13].
EAC cells were collected from Chittaranjan National Cancer Institute (CNCI), Kolkata, India. The EAC cells were maintained in Swiss albino mice, by intraperitoneal transplantation on every 9th days [14] .The ascitic fluid was collected from a donor mouse by syringe and the tumor cell count was performed under microscope in a Neubauer haemocytometer manufactured by Marienfield, Germany. 2 X 107 cells /ml were obtained by dilution with normal saline [15] .Tumor cell suspension showing 98% viability (checked by trypan blue dye (0.4%) exclusion assay) was used for transplantation. A definite number (about 2 x 106 / 0.2mL) of these living viable cells was injected or implanted into the peritoneal cavity of each mouse in 3 groups except normal saline group (negative control). In this instance, the tumor cells multiplied relatively freely within the peritoneal cavity and ascites developed. This was taken as Day 0. A day of incubation was allowed to establish the disease in the body before starting the drug administration. All the animals were thus induced tumor by injecting EAC cells to peritoneal cavity. The EAC induced mice as divided into 3 groups (n = 6), were Pure 5-FU treated, Formulation ‘FF5’ loaded with 5-FU treated, and no treatment(positive control). The treatment groups were administered 20 mg/kg /day of the drug (5-FU) intravenously through tail vein or its equivalent amount (in the case of formulation ‘FF5’) in the transdermal patch applied on the dorsal surface of mice for 9 consecutive days. The control group was kept as untreated [16].
Group I: 0.9% Normal saline (5ml/kg i.p) Negative Control
Group II: EAC (2X106 cells/mouse i.p.) Positive Control
Group III: EAC (2x106 cells/mouse i.p.) +Pure 5-fluorouracil (20mg/kg/day) intravenously through tail vein.
Group IV: EAC (2x106 cells/mouse i.p.) + Formulation ‘FF5’ (equivalent to 5-FU 20mg /kg /day) transdermal patch applied on the dorsal surface.
The major problem encountered was how to protect the applied transdermal film from being scratched off, rubbed off and/or licked off, during the experimental procedure, once applied to the shaved dorsal surface of the skin of the mice. The Velcro protection jacket was designed, with minor modifications according to the description of [17]. The Velcro jacket was made to cover the entire trunk of the mice and open at the top, which was appropriately designated for application of transdermal film. The jacket protected the transdermal film and allowed for good ventilation. It served its purpose quite well and the mice were able to function normally while wearing it. Antitumor activity of formulation ‘FF5’ was assessed by observing the changes with respect to the following parameters. On the tenth day food and water was withheld 6 hr before the starting of the testing operation.
The effect of formulation ‘FF5’ loaded with 5-FU on tumor growth was examined by studying the following parameters such as tumor volume, packed cell volume, tumor cell count, viable tumor cell count, nonviable tumor cell count and percentage increase in life span.
The mice were dissected and the ascitic fluid was collected from the peritoneal cavity by a syringe. The volume was measured by taking it in a graduated centrifuge tube. Packed cell volume was determined by centrifuging the ascitic fluid at 1000 rpm for 5 minutes at 22-25°C in a cold centrifuge (Remi).
The ascitic fluid was taken in a WBC pipette and diluted to 100 times. Then a drop of the diluted cell suspension was placed on the Neubauer counting chamber and the number of cells in the 64 small squares was counted carefully.
The cells were then stained with trypan blue (0.4% in normal saline) dye. The cells that did not take up the dye were viable and those which took the stain were nonviable. These viable and nonviable cells were counted. Comparing the tumor volume, packed cell volume and viable cell count of the test with that of control made the evaluation of the test sample. The percentage inhibition of tumor volume and viable cell count of the ascitic fluid was obtained by the following expression:
Percentage inhibition of Ascitic fluid = (1 – T / C) x 100
Where, T = Average volume (ml) of Ascitic fluid in test animals, C = Average volume (ml) of Ascitic fluid in control animals and then
Percentage inhibition of Ascitic cells = (1 – T / C) x 100
T= Average no. of Ascitic cells in test animals and C° = Average no. of Ascitic cells in control animals.
The effect of formulation’FF5’ on tumor growth was also monitored by recording the mortality daily for 6 weeks and percentage increase in life span (%IMST) was calculated. An enhancement of life span by 25% or more was considered as effective antitumour response [13,18].
IMST (%)=[(Median survival time of treated group/ Median survival time of control group)- 1]X100
Median Survival Time (MST) = [Day of first death + Day of last death]/ 2
Values were presented as mean ± S.E.M.Data were statistically evaluated by one-way analysis of variance (ANOVA) followed by post hoc Dunnett’s test using SPSS software. P< 0.05 was considered as statistically significant and p< 0.01 as highly significant.
Percentage inhibition of Ascitic fluid: = (1 – T / C) x 100
Where, T = Average volume (ml) of Ascitic fluid in test animals, C = Average volume (ml) of Ascitic fluid in control animals :
(i) Pure drug (5-FU) == 65.05% compared to control.
(ii) Formulation ’FF5’ == 84.17% compared to control. Similarly,
(i) Pure drug (5-FU) ==72.88%
(ii) Formulation’FF5’ ==79.11%
and then
Percentage inhibition of Ascitic cells: = (1 – T/ C) x 100
T = Average no. of Ascitic cells in test animals and C° = Average no. of Ascitic cells in control animals:
(i) Pure drug (5-FU) == 70.31% compared to control.
(ii) Formulation ’FF5’ == 88.71% compared to control.
The above in -vivo studies indicates that Antitumor activity of transdermal film 5-FU (FF5) reduces tumor incidence in mice by 84.17% with respect to Ascitic fluid, (79.11%) of packed cell volume and 88.71% with respect to Ascitic cells when the pure drug (5-FU) reduces 60.05% of ascitic fluid, (72.88%) of packed cell volume and 70.31% of Ascitic cells, compared to EAC induced control mice.
Cancer is a pathological state involving uncontrolled proliferation of tumor cells.
The present study was carried out to investigate the antitumor potential of our prepared formulation ’FF5’ loaded with 5-FU against EAC bearing mice. EAC (Ehrlich Ascites Carcinoma) is a very rapidly growing carcinoma with very aggressive behavior [19]. It is able to grow in almost all strains of mice. The Ehrlich Ascitic tumor implantation induces per se a local inflammatory reaction, with increasing vascular permeability, which results in an intense edema formation, cellular migration, and a progressive ascitic fluid formation and accumulation [20] .
The ascitic fluid is essential for tumor growth, since it constitutes a direct nutritional source for tumor cells [21] . Our Formulation ‘FF5’ loaded with the drug treatment significantly (p<0.01) reduced tumor volume probably by lowering the ascitic nutritional fluid volume. Further, the packed cell volume and the number of viable EAC tumor cells in peritoneum were significantly reduced in the mice treated with the formulation ‘FF5’ when compared to the tumor control group. These results could indicate either a direct cytotoxic effect of formulation ’FF5’ on tumor cells or an indirect local effect, which may involve macrophage activation and vascular permeability inhibition.
The reliable criterion for judging the value of any anticancer formulation is the prolongation of life span of the animal [22] .The increase in the life span of tumor bearing mice treated with ‘FF5’ film and pure drug 5-FU was found to be 88.95% and 68.80% respectively as compared to the control groups (table-10). The ‘FF5’ decreased the ascites fluid volume and thereby increased the percentage of life span.
From the stability study data we observe that the formulations are stable in accelerated as well as intermediate conditions. Hence, the above in- vivo investigation showed highly significant inhibition of tumor volume, packed cell volume and viable tumor cell count with the simultaneous increase in percentage of non-viable cell count in EAC bearing mice (table-9). Thus the formulation ’FF5’ contributes a positive response and supports the antitumor effect of formulated film.