ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
Samy M. El-Megharbela,b*, Ahmed El-Maghrabya,c and Moamen S. Refata,d
|
| Corresponding author:Prof. Moamen S. Refat |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Mn(II), Co(II), Zn(II) and Cd(II) carbonates, respectively, were achieved by a new chemical procedure under the reaction of aqueous solutions of metal ions (MnCl2, Co(NO3)2, Zn(NO3)2 or CdCl2), potassium iodide and urea with molar ratio 1:2:10, respectively, at ~ 90 oC for 3 hrs. The infrared spectra of the results indicate absence of the essential bands of urea, but existed of the characteristic bands of ionic carbonate, CO3 2–. An important chemical mechanisms discussed the preparation of Mn(II), Co(II), Zn(II) and Cd(II) carbonate compounds were suggested.
Keywords |
| carbonate CO3 2–, infrared spectra, transition metals, potassium iodide, urea. |
INTRODUCTION |
| Urea is physiologically very important. It is the chief nitrogenous product of protein metabolism. Urea has a melting point of 132&C, soluble in water and ethanol, but insoluble in ether. Urea is used for preparing formaldehyde- Urea resin (plastics) [1], barbiturates [2], and fertilizers [3-6]. Urea is also extensively used in the paper industry to soften cellulose and has been used to promote healing in infected wounds and many other applications in the field of biological and organic synthesis [7-10]. Yamaguchi and Stewart [11, 12] were assigned all of the observed frequencies in the spectra of urea and urea-d4. The two vibrations of the frequencies at 1686 and 1603cm-1 were assigned as the 1686 cm-1 band due to CO stretching vibration and the 1603 cm-1 band for NH2 bending motion. The calculations studied by Yamaguchi showed that for the band at 1686 cm-1, the contribution of the NH2 bending motion is greater than that of CO stretching motion. The infrared bands of urea-d4 observed at 1245 and 1154 cm-1 are assigned to ND2 bending vibrations. This assignment is consistent with the observed depolarization degrees of the Raman lines. The 1464cm-1 frequency of urea is assigned to the CN stretching vibration. The corresponding frequency of urea-d4 is observed at 1490cm-1. The 1150cm-1 band is assigned to NH2 rocking vibrations. The reactions between transition metal ions and urea at room temperature have been studied extensively [13-17]. The infrared spectra of these complexes clearly indicated that urea molecule behaves as a mono dentate ligand and coordinates to the metal ions through the oxygen atom and not the nitrogen atom. The nature of the reaction products depend strongly on the type of metal ions and so the metal salt used. The novelty in our previously studies [18-27] were oriented to the reaction of urea ligand with different metals such as Co (II), pb(II), Sn(II), Cr(III), Fe(III), Au(III), Sn(IV), V(V) and Mo(IV) at high temperature which demonstrate that the types of metal ions beside their anions have a pronounced effect on the nature of the reaction products. The published papers were trended for the reaction of urea with different metal salts at elevated temperature lead to discovering a novel method for preparation pbCO3 and CoCO3 [21], lanthanide carbonates [23,27], limonite, FeO(OH) [20], 2ZnCO3.3Zn(OH)2 [19], SnOCl2.2H2O [18], (Cr2O3, MnO2, MoO3 and WO3) oxides resulted from a novel oxidation reduction reaction between (K2CrO4 or K2Cr2O7), KMnO4, Na2MoO4 and Na2WO4, respectively, with urea in an aqueous solution at ~ 85 oC [27]. The main task in this paper was undertaken to identify the nature of the reaction mechanisms of the products resulted during the reaction of (MnCl2, Co(NO3)2, Zn(NO3)2 or CdCl2), potassium iodide and urea with molar ratio 1:2:10, respectively, at ~ 90 oC for 3 hrs in aqueous media. The reaction products were isolated as solids and characterized by infrared spectroscopy technique. |
II. EXPERIMENTAL |
| All chemicals used throughout this work were analytical pure. MCO3 (M = Mn(II), Co(II), Zn(II) or Cd(II)) were prepared by mixing an aqueous solutions (100 ml) of 0.1M of urea, 0.02 of potassium iodide with 0.01M of the respective MnCl2, Co(NO3)2, Zn(NO3)2 or CdCl2. The mixtures were heated at 90 oC for 3 hrs in a hot plate. The solid products compounds were filtered off, washed several times with hot water, dried at 80 oC in an oven for 3 hours and then placed in vacuo over anhydrous calcium chloride. The yields of the obtained Mn(II), Co(II), Zn(II) and Cd(II) carbonates were varied in the range 70-to-80% depending upon the type of metal as well as on the counter ions associated with the metal ion. Carbonate content in the four compounds were determined by dissolving a sample of each product in excess standard HCl and the excess of HCl was determined using standard sodium carbonate [28]. The percentage of manganese(II), cobalt(II), zinc(II) and cadmium(II) in their compounds were determined gravimetrically method till constant weight and stable formula. The infrared spectra of urea, all reactants and products were recorded using a Bruker FT-IR Spectrophotometer. |
III. RESULTS AND DISCUSSION |
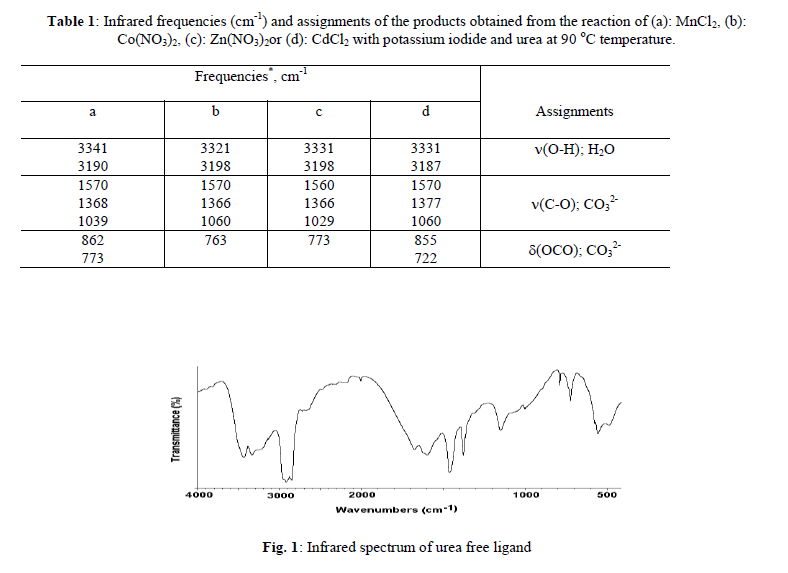
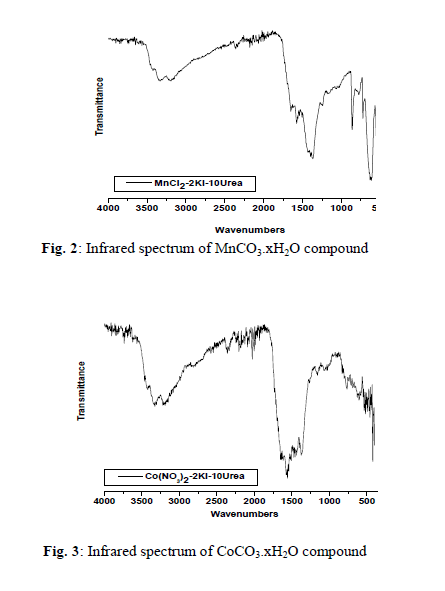
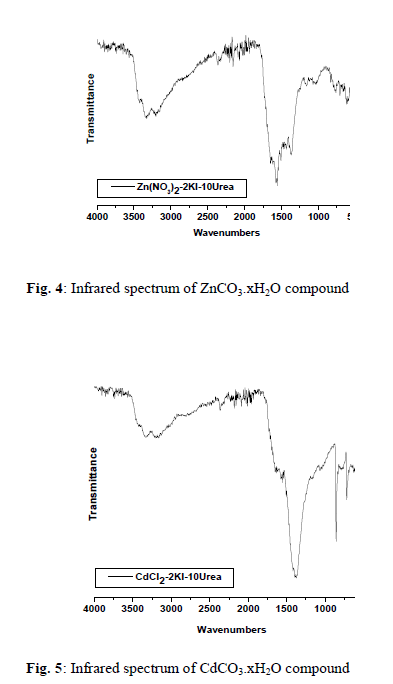
| The chemical reactions of 0.1M of urea, 0.02 of potassium iodide with 0.01M of the respective MnCl2, Co(NO3)2, Zn(NO3)2 or CdCl2 with molar ratio (10: 2: 1), respectively, at 90 o C produces a buff, violet, white and white solid fine powders, respectively. The infrared spectra of urea as well as the reaction products of different manganese(II), cobalt(II), zinc(II) and cadmium(II) salts with urea at low temperature (90 o C) were obtained. The spectra of free urea ligand, manganese(II), cobalt(II), zinc(II) and cadmium(II) carbonates are shown in Fig. 1-5, respectively. The band assignments for the products are given in Table 1. The infrared spectra show no bands due to any of the reactants and of coordinated urea, but instead, a group of bands characteristic for the ionic Carbonate, (CO3)2-, is appeared [29]. Based on this fact, along with that obtained data from the volumetric determination of (CO3)2- group with standard solution of HCl and beside that the infrared spectra of the commercially obtained MnCO3, CoCO3, ZnCO3 and CdCO3 are the same as that of the reaction products. The products obtained were identified as MnCO3, CoCO3, ZnCO3 and CdCO3. The infrared assignments agree quite well with those known [29] for the ionic carbonate (CO3)2-. Previous studies [18-27] indicated that the nature of the reaction product obtained from the reaction of metal ions with urea at high temperature depends upon the type of metal ion and in some cases on the nature of the metal salts used. The manganese(II)-urea complexes have been studied extensively [30-33] and it was found that, these complexes are of high spin and possess octahedral structures. These investigation have also shown that, all of the studied Mn(II)- urea complexes are of the general formula MnU2X2 (X=Cl– or Br–, U=Urea). They have a chain like structure with halogen bridges. This structure is supported by electronic spectral measurements and the magnetic susceptibility data [34] but could not be checked by infrared spectroscopy because of the Mn-X-M bridging vibration frequency lying below 200 cm-1.The trans-octahedral complex [MnU4(NCS)2], (U=urea) has been prepared and investigated [35]. The ligand, urea is coordinated through its oxygen atom while the thiocyanate through the nitrogen atom. These results are supported by the crystallographic data of Tsintsadze et al [36] obtained or the [MnU4(NCS)2] complex. The trifluorobis(urea)manganese(III) trihydrate, [MnF3(urea)2].3H2O complex is prepared and its infrared spectrum is obtained [37]. The characteristic features of the infrared spectrum of this complex are the absorptions due to coordinated urea, coordinated fluoride and lattice water. The important bands of coordinated urea have been observed at 3425 and 1535cm-1, assignable to (NH2) and (CO) modes, respectively. The unaltered position of (NH2) and a significant lowering of (C=O) band frequencies compared with those for the free urea suggest that urea is coordinated with the metal in an unidentate manner through its oxygen atom. However, there is a shift to the lower frequency in the position of the (Mn-F) mode indicating that, the fluoro ligands are involved also in the formation of bridges among the contiguous Mn centers in the crystal lattice. At high temperature the role of Mn(II) ions in decomposing the coordinated urea in the form of [MnU2I2] could be understood according to the following reactions; |
 |
| 2, 3 and 4 are infrared active. The stretching vibrations of the type; (C-O); 3(E`) is observed as a strong broad band in the range of 1550-1360 cm-1 while the stretching vibration (C-O); 1(A`1) is observed in the region ~1030 cm-1 as a medium-to-weak band. It should be indicated here that this motion (A`1) should be infrared inactive, its weak appearance in the spectrum of MCO3 could be related to weak solid-solid interactions causes the symmetry of CO3 2- to be lowered than D3h. The out of plane of vibration (OCO); 2(A``2) is observed in the range of ~800 cm-1 as a strong band while, the angle deformation bending vibration (OCO); 4(E`) appear in the range of 700 cm-1 as a medium strong. The infrared spectra of metal(II) carbonate, MCO3 show that, some these products are hygroscopic and its clearly have moisture water. The band related to the stretching vibration (O-H) of uncoordinated H2O is observed as expected in the range of ~ 3000 cm-1 Table 1. |
 |
 |
 |
References |
|