ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
A.H.Rajanna1, S.L.Belagali2
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Environmental contamination due to persistence of pesticide residues is gaining worldwide attention. Exposure to pesticide residues can represent a potential risk to any organisms. The residue level of pesticides were quantified in 16 samples during the year 2013, among those four for water, five for sediment samples from Kabini river, three for ground water and four for agricultural soil samples from the Nanjangud region of Southern India. Samples were extracted for pesticide preparation prior to analysis with Gas Chromatography. A total of 12 Organochlorine pesticides were examined. The experimental results revealed that, isomers of HCH, DDT and Heptachlor were detected in water and soil samples, whereas, β-isomer was detected in the sediment samples. Among the isomers of HCH, β- isomer was predominant in all the studied samples. Detection of pesticide residues shows serious threat to the ecosystem. Thus, water and soils in the vicinity of the industries need to be paid much attention
Keywords |
| Pesticides, Nanjangud, Kabini river, Soil. |
INTRODUCTION |
| Modern agriculture is readily associated with the continuous and indiscriminate use of different agricultural chemicals or pesticides, which may not only harm the indigenous soil microorganisms but also disturb the soil ecosystem and soil health [1]. Growing demand for food as a result of increasing population has led to substantial rise in the production and utilization of a wide variety of pesticides in agriculture [2]. The amount of applied pesticides reaching the target organisms is about 0.1%, while the remaining bulk contaminates the soil environment [3]. Extensive use of these chemicals has given rise to several short-term and long-term adverse effects. It has been observed that, their long-term, low-dose exposure increasingly link to human health effects such as immune-suppression, hormone disruption, diminished intelligence, reproductive abnormalities and cancer [4]. Organochlories are often distributed heterogeneously in the environment and hence, exposure likely spatially explicit [5]. Organochlorine compounds such as PCBs, DDTs and HCH are among the most widely known class of contaminants, because of their ubiquity, potential for magnification in the food chain and harmful biological effects [6]. The constant usage of pesticides leads to a continued contamination of the environment including water resources [7], [8], [9], [10]. Organochlorine pesticides are still used extensively in India due to their effectiveness and low cost [11], [12]. The water supplies of several Indian cities as well as the ground water in rural areas have been contaminated with high levels of pesticides, mainly the organochlorines [13], [14]. Organochlorine pesticides are known to resist degradation and therefore can be redistributed through the food chain and produce significant contamination at the apex of the food chain [15]. No work has been directed towards the contamination of persistent pesticides in Nanjangud area. Thus, the present study aims to identification and quantification of organochlorine compounds in agricultural soil, Kabini river water and sediment samples in the Nanjangud region of southern India. |
BACKGROUND OF THE STUDY AREA |
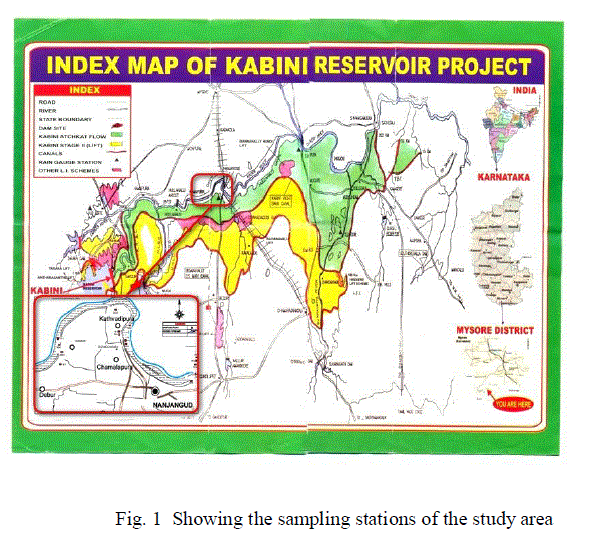 |
| Nanjangud is one of the most important industrial places situated near Mysore in the State of Karnataka. Nanjangud is spread over from 12º 06′ 39″ N longitude to 76º 33′ 46″ E latitude and 12º 07′ 56″ N longitude to 76º 42′ 9″ E latitude. It as an average elevation of 657 meters. The total geographical area of Nanjangud is 991 sq. km. According to the 2001 census, the total population of the talk was 3,60,223 and annual average rainfall was 670 mm. The river Kabini takes its origin in the Western Ghats in Wyanadu district of Kerala state and flows for a length of 230 km before joining the river Cauvery at Tirumakudalu in Karnataka state. The river flow system irrigates 44,516 hectares of agricultural field, out of which 21,448 hectares are for paddy and 23,068 hectares of semi-dry crops. Red loamy soil and sandy loam represent the major soil type of the study area. The Kabini river is bordered on both sides by paddy fields and the river water is much used for irrigation purpose. |
MATERIALS AND METHODS |
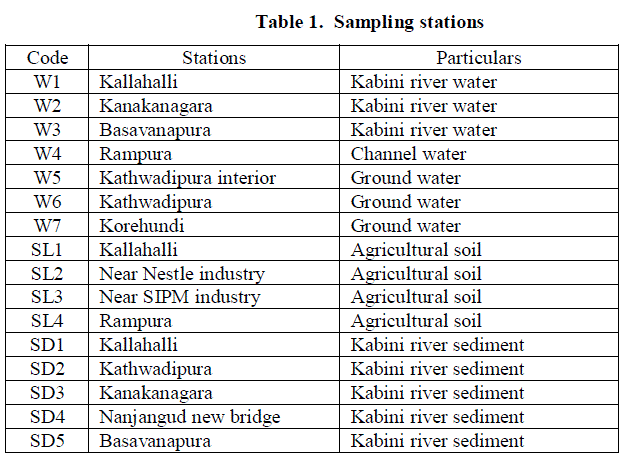
| Sampling stations were selected, keeping in view, the important industrial waste discharge zones and nature of domestic activities and agricultural run-off. As shown in the (Figure 1), water samples were collected from seven sites. Out of seven, three sites for Kabini river water (W1, W2 and W3), one site (W4) for channel water and another three sites (W5, W6 and W7) for bore well water. Soil and sediment samples were collected from nine sites. Out of nine sites, four for agricultural soil (SL1, SL2, SL3 and SL4) and five for Kabini river sediments (SD1, SD2, SD3, SD4 and SD5). Particulars of the sampling stations were given (Table 1). |
 |
| For the analysis of pesticide residues, water samples were collected in one liter amber colored bottles transferred to a separating funnel. Sodium sulphate (10 g) was added to the separating funnel and shaken well. The water-sodium sulphate mixture was extracted with dichloromethane (3x100 ml). After each separation, the upper organic layer was collected in a separate beaker and the lower aqueous layer was again extracted with 100 ml of dichloromethane. The combined dichloromethane layers were reduced in volume on a rotary evaporator to about 5 ml. The crude extracts were cleaned up by florisil column chromatography. Glass columns were packed from the bottom with a glass wool plug, 8 cm of deactivated florisil and 4 cm anhydrous Na2SO4 to remove excess oil and fat and moisture content respectively. The packed column was pre-washed with 50 ml of petroleum ether. The extract was transferred to the column and eluted with 200 ml petroleum ether, mixed with diethyl ether (85:15). The combined extracts were evaporated almost to dryness and the final volume reached to 5 ml with GC grade solvent as described in APHA [16] standard methods and the water extracts were subjected to GC analysis. Composite sampling of agricultural soil samples were collected in polythene bags from the depths of 10-15 cm employing tube auger sampler, from each of the selected sampling points. Sediment samples were collected from the Kabini river bed employing an Ekman dredge sampler. For the extraction of soil samples for determination of pesticides, 50 g of each soil and sediment samples was dried in a natural environment, weighed and added with ammonium chloride solution (NH4Cl), followed by acetone and shaken well for 20 minutes. Then 50 ml petroleum ether was added and kept on a shaker for overnight. After filtering by Whatman No. 1 filter paper, 50 ml of acetone was added to each soil sample and was filtered again. To remove excess solvents, Rotary Evaporator was used. The crude extracts were cleaned up by florosil column chromatography, to remove excess oil, fat and moisture contents. The packed column was pre-washed with 50 ml of petroleum ether. The extracts were transformed to the column eluted with 160 ml petroleum ether mixed with diethyl ether. The combined extracts were evaporated almost to dryness and the final volume of 5 ml was obtained with GC grade solvents. The identification and quantification of HCH, Aldrin and DDT compounds were carried out using a Gas Chromatography (Chemito GC 1000), with Electron Capture Detector. GC column employed were capillary column, DB-1701 and DB-5. Pure analytical grade pesticide standards were used for GC analysis as reference standards. The temperature programs of GC were: injector 230o C, column 220oC and detector 260o C. Ultra pure nitrogen gas was used as carrier gas. Winchrome 1000 chromatography data processor was used to record the chromatograms and peak areas were used to calculate the pesticide residues in the samples. These compounds were identified as individually resolved peaks based on retention times, in comparison with the corresponding peak height of the standards. |
RESULTS AND DISCUSSION |
 |
 |
 |
 |
 |
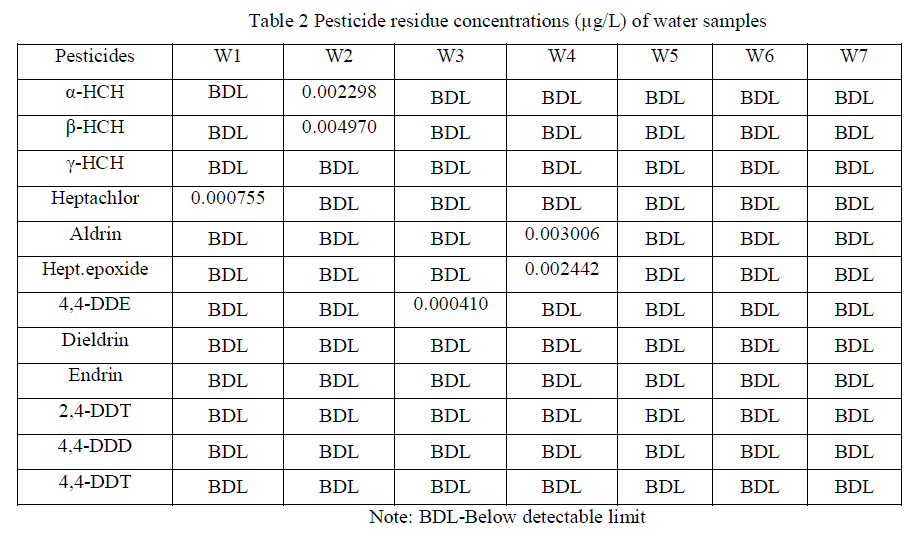
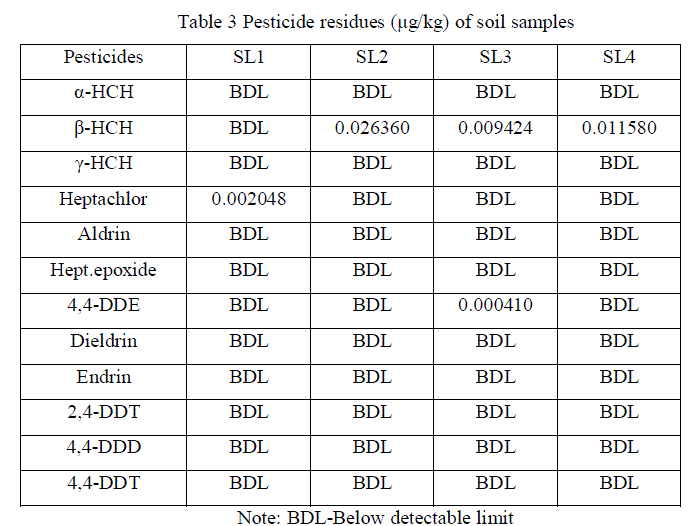
| Detection of pesticide residues in the water samples are summarized (Table 2) and Residue levels of organochlorines were represented (Figures 2, 3 and 4). It shows that, organochlorine residues in the water samples are Heptachlor (0.000755 μg/L) for W1 sample, α-HCH (0.002298 μg/L) & β-HCH (0.004970 μg/L) for W2 sample, 4,4'-DDE (0.000410 μg/L) for W3 sample and Aldrin (0.003006 μg/L) & Heptachlor epoxide (0.002442 μg/L) for W4 sample. Although DDT was not detected in any of the samples, one of its metabolites, 4,4'-DDE was detected. 4,4-DDE found in the environment might have resulted from the pesticide application, other than agricultural uses, especially for vectors [17]. Hexachlorocyclohexane (HCH) used for locust control, Heptachlor used for control soil inhibiting pests, Aldrin was effective against wireworms and to control termites [4]. The β-isomer of HCH has a higher aqueous solubility than either the α- or γ-isomers which could account for the high levels in aquatic ecosystems, following drainage and runoff from agricultural regions [18]. Pesticide residues were not detected in any of the ground water samples. Further, runoff from agricultural fields and washing of the spraying equipments in the water body are also responsible for detection of pesticide residues in the surface water. Detection of pesticide residues in the soil samples are summarized (Table 3). It reveals, Heptachlor (0.002048 μg/kg) for SL1 sample, β-HCH (0.026360 μg/kg) for SL2 sample, β-HCH (0.009424 μg/kg) & 4,4'-DDE (0.000410 μg/kg for SL3 sample, β-HCH (0.011580 μg/kg) for SL4 sample. Detected residues might be due to the application of insecticides, herbicides, fungicides and rodenticides to prevent diseases attack of the crops. Detection of pesticide residues in the sediment samples are summarized (Table 4) and reveals that, β-HCH were detected in the concentration of 0.009194 μg/kg for SD1, 0.05086 μg/kg for SD3, 0.03640 μg/kg for SD4 and 0.02528 μg/kg for SD5 samples respectively and no pesticide residues were detected in SD2 sample. Detected residues might be due to the runoff of residues from agricultural fields and application for other than non agricultural uses, especially vectors are also responsible for accumulation of residues in the river sediments. Among the isomers of HCH, β-isomer was predominant in soil and water followed by α-and γ-isomers, whereas, in sediment samples only β-isomer was detected. High concentration of β-HCH might be due to the isomerization of γ- HCH to – β-HCH in water and sediment as suggested by Hayes (1982). The technical composition is mainly composed of α-(67%), β-(10%), γ-(15%), δ-(8%). Among all the HCH isomers, β-HCH has the lowest vapour pressure, is persistent in nature and tends to accumulate in different soils [19]. Therefore, the concentration of this isomer in the studied samples was likely to remain at a high level. |
CONCLUSION |
| The indiscriminate use of pesticides has increasingly become a matter of environmental concern as it accumulates in the soil. Among the isomers of HCH, β-isomer was predominant in soil and water, based on the experimental results it can be concluded that, detection of pesticide residues in both soil and river water samples, which might be due to the application of pesticide residues to boost up of the crop yields and application for other than non agricultural uses, especially for vectors are also responsible for the accumulation of residues in the river system. Moderen agricultural practices and effluent released from the industries are degrading the environmental quality of Kabini river and agricultural soil in the vicinity of Nanjangud industrial area. Thus, water and soils in the surroundings of the industries need to be paid much attension. |
ACKNOWLEDGEMENT |
| The author aknowledge the assistant of Karnataka state pollution control board for providing necessory equipment facility for pesticide analysis. |
References |
|