ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
| Gurjaspreet Singh Department of Chemistry, Panjab University, Chandigarh, India |
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
This manuscript deal with the reaction of the 1-Isothiocyanato six membered silatrane with different Lewis acids. The adducts 1, 2, 3 and 4 so formed were characterized by elemental analysis, infrared spectroscopy, 1H, 13C, 29Si NMR spectroscopy. η1-S and η1-N bonding pattern were observed the coordination modes of the isothiocyanato group
KEYWORDS |
| Lewis acid, silatrane, isothiocyanato |
I. INTRODUCTION |
| The coordination behaviour of organic isothiocyanato has been extensively studied in the past [1-4]. The isothiocyanato group exhibits a variety of coordination modes depending upon the nature of Lewis acid used in most of the known coordination compounds of this class [5]. The results have been amply rationalized on the basis of hard/soft acid-base character (in Pearson’s sense) of nitrogen as well as sulphur site respectively amongst the wide variety of coordination modes of isothiocyanato group. The η1-N, η1-S bonding pattern are found to be the most common in the absence of Xray crystal data. IR spectroscopy has remained the most versatile tool to identify these coordination modes [6-9]. |
| In contrast to the organic isothiocyanato, little is known about the coordination behavior of silyl isothiocyanates partially due to the fragile nature of Si-N bond in these compounds [6,10-11]. It is in this context that the reactivity of 1-Isothiocyanato six membered towards Lewis acids has been undertaken. |
II. EXPERIMENTAL |
| All operations were carried out under dry nitrogen atmosphere. Solvents were freshly distilled under inert atmosphere from sodium (hexane), and phosphorus pentaoxide (dichloromethane) before use. Hydrous iron(III) chloride, nickel(II) chloride, cobalt(II) chloride, copper(II) chloride were converted to their anhydrous form using thionyl chloride. 1- Isothiocyanato six membered ring silatrane was synthesized according to procedure reported in literature [6]. IR spectra were obtained as thin films or nujol mulls on Perkin-Elmer RX-1 FTIR spectrophotometer. 1H (300.4 MHz), 13C (75.45 MHz), 29Si NMR (59.60 MHz) spectra were obtained on JEOL AL 300 instrument. Chemical shifts were reported with respect to TMS as an external standard. |
III. SYNTHESIS |
Reaction of of 1-Isothiocyanato six membered silatrane and iron(III) chloride (1) |
| 1-Isothiocyanato six-membered ring silatrane (1.00 g, 1.99 mmol) was dissolved in dry dichloromethane in a twonecked round-bottomed flask. The solution was stirred with the help of magnetic stirrer. Iron(III) chloride (0.32 g, 1.99 mmol) was added to the solution of 1-Isothiocyanato six-membered ring silatrane. A dark brownish black solid was precipitated out. The reaction mixture was stirred at room temperature for 4 h. The solid thus obtained was filtered washed with n-hexane and dried under reduced pressure. |
Reaction of of 1-Isothiocyanato six membered silatrane and cobalt(II) chloride (2) |
| 1-Isothiocyanato six-membered ring silatrane (1.00 g, 1.99 mmol) was dissolved in dry dichloromethane in a twonecked round-bottomed flask. The solution was stirred with the help of magnetic stirrer. Cobalt(II) chloride (0.25 g, 1.99 mmol) was added to the solution of 1-Isothiocyanato six-membered ring silatrane. A white solid immediately precipitated out. The reaction mixture was stirred at room temperature for 4 h. The solid, thus obtained was filtered washed with n-hexane and dried under reduced pressure. |
Reaction of of 1-Isothiocyanato six membered silatrane and nickel(II) chloride (3) |
| 1-Isothiocyanato six-membered ring silatrane (1.00 g, 1.99 mmol) was dissolved in dry dichloromethane in a twonecked round-bottomed flask. The solution was stirred with the help of magnetic stirrer. Nickel(II) chloride (0.25 g, 1.99 mmol) was added to the solution of 1-Isothiocyanato six-membered ring silatrane. A yellowish white solid immediately precipitated out. The reaction mixture was stirred at room temperature for 4 h. The solid thus obtained, was filtered washed with n-hexane and dried under reduced pressure. |
Reaction of of 1-Isothiocyanato six membered silatrane and copper(II) chloride (4) |
| 1-Isothiocyanato six-membered ring silatrane (1.00 g, 1.99 mmol) was dissolved in dry dichloromethane in a twonecked round-bottomed flask. The solution was stirred with the help of magnetic stirrer. Copper(II) chloride (0.26 g, 1.99 mmol) was added to the solution of 1-Isothiocyanato six-membered ring silatrane. A brownish black solid immediately precipitated out. The reaction mixture was stirred at room temperature for 4 h. The solid thus obtained was filtered washed with n-hexane and dried under reduced pressure. |
IV. RESULTS AND DISCUSSION |
| a) Reaction of 1-Isothiocyanatosilatrane possessing six membered ring with iron(III) chloride The reaction of equimolar quantities of 1-Isothiocyanato six membered silatrane and iron(III) chloride in dichloromethane medium at room temperature (25 ïÃâðC) results in the formation of a dark brown solid. Elemental analysis of this product corresponds to the composition FeCl3.NCSSi[(OC6H2Me2)CH2]3N. The isolated compound is hygroscopic in nature and insoluble in the most of the solvents except dimethyl sulfoxide. The reaction may be represented as follows (Scheme 1): |
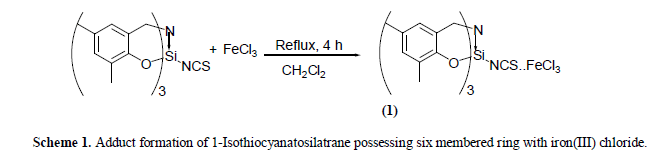 |
 |
| NMR Spectra: Though, DMSO-d6 is suspected to dissociate the complex, it has been used as a solvent to study the multinuclei 1H, 13C and 29Si NMR spectra, since the adduct is insoluble in most of the other solvents. 1H NMR spectrum of the adduct 1 in DMSO-d6 reveals two singlets of equal intensity at 2.18 and 2.24 ppm due to aromatic methyl protons and singlet at 4.22 ppm due to NCH2, two singlets of equal intensity at 6.85 and 6.89 ppm due to aromatic protons. |
| 13C NMR spectrum of the adduct 1 in DMSO-d6 reveals two singlets at 15.97 and 19.79 ppm due to aromatic methyl carbons, singlet at 55.12 ppm due to NCH2. The carbon atom of the isothiocyanato group appears as a triplet at 127.13, 127.32 & 127.48 ppm due to13C-14N coupling [1J (13C-14N=13.20Hz)] and singlets at 117.1, 125.18, 129.11, 131.10 and 150.96 ppm due to aromatic carbons. |
| Similarly, the 29Si NMR spectrum of the complex reveals a triplet of equal intensity at -145.13, -145.72 and -146.25 ppm with a coupling constant [1J (29Si-14N) = 33.04 Hz]. Apparently, the chemical shifts are identical to those of pure silatrane. Thus, on the basis of identical NMR chemical shift with respect to parent silatrane, it may be concluded that the adduct dissociates in DMSO-d6. NMR studies are quite informative since these provide irrevocable evidences in favour of retention of Si-NCS bond even in the presence of strong Lewis acid (FeCl3). |
| b) Reaction of 1-Isothiocyanatosilatrane possessing six membered ring with cobalt(II) chloride |
| The reaction of equimolar quantities of 1-Isothiocyanato six membered silatrane and cobalt(II) chloride in dichloromethane medium at room temperature (25 ïÃâðC) results in the formation of a white solid. Elemental analysis of this product corresponds to the composition CoCl2.NCSSi[(OC6H2Me2)CH2]3N. The compound isolated above is hygroscopic in nature and insoluble in the most of the solvents but soluble in dimethyl sulfoxide.The reaction may be represented as follows (Scheme 2): |
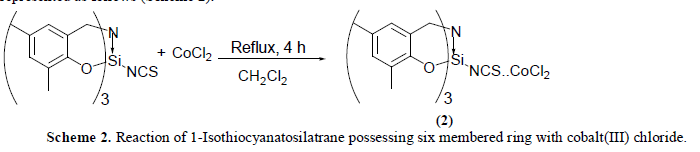 |
 |
| NMR Spectra: The poor solubility of the adduct in non-coordinating solvents has precluded detailed NMR studies of the adduct. However, the multinuclei (1H, 13C and 29Si) NMR spectra in DMSO-d6 solvent have been recorded in order to gather evidences in support of retention of silatrane moiety in the adduct. The results are described below. 1H NMR spectrum of the adduct 2 in DMSO-d6 reveals two singlets of equal intensity at 2.21 and 2.23 ppm due to aromatic methyl protons and singlet 4.25 ppm due to NCH2, two singlets of equal intensity at 6.89 and 6.92 ppm due to aromatic protons. |
| 13C NMR spectrum of the adduct 2 in DMSO-d6 reveals two singlets at 16.11 and 19.82 ppm due to aromatic methyl carbons, singlet at 55.10 ppm due to NCH2 The carbon atom of the isothiocyanato group appears as a triplet at 128.53, 128.72 & 128.87 ppm due to13C-14N coupling [1J (13C-14N=12.82Hz)] and singlets at 116.41, 124.28, 129.02, 129.44 and 150.96 ppm due to aromatic carbons. Similarly, the 29Si NMR spectrum of the complex reveals a triplet of equal intensity at –149.28, -149.86 and -150.41 ppm with the coupling constant [1J (29Si-14N=33.33 Hz)]. Apparently, the chemical shifts are identical to those of pure silatrane. |
| c) Reaction of 1-Isothiocyanatosilatrane possessing six membered ring with nickel(II) chloride |
| The reaction of equimolar quantities of 1-Isothiocyanato six membered silatrane and nickel(II) chloride in dichloromethane medium at room temperature (25 ïÃâðC) results in the formation of an yellowish white solid. Elemental analysis of this product corresponds to the composition NiCl2.SCNSi[(OC6H2Me2)CH2]3N. The compound isolated above is hygroscopic in nature and is insoluble in most of the solvents except dimethyl sulfoxide. The reaction may be represented as follows (Scheme 3): |
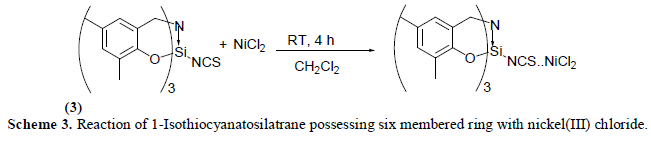 |
 |
| NMR Spectra: Though, DMSO-d6 is suspected to dissociate the complex, it has been used as a solvent to study the multinuclei 1H, 13C and 29Si NMR spectra, since the adduct is insoluble in most of the other solvents. 1H NMR spectrum of the adduct 3 in DMSO-d6 reveals two singlets of equal intensity at 2.19 and 2.20 ppm due to aromatic methyl protons and a singlet at 4.25 ppm due to NCH2, two singlets of equal intensity at 6.89 and 6.91 ppm due to aromatic protons respectively. |
| 13C NMR spectrum of the adduct in DMSO-d6 reveals two singlets at 16.10 and 19.11 ppm due to aromatic methyl carbons, singlet at 54.99 ppm due to NCH2. The carbon atom of the isothiocyanato group appears as a triplet at 127.69, 127.89 &128.04 ppm due to 13C-14N coupling [1J (13C-14N=13.20Hz)] and singlets at 116.47, 124.38, 129.23, 129.51 and 150.96 ppm due to aromatic carbons. |
| Similarly, the 29SiNMR spectrum of the complex reveals a triplet of equal intensity at –151.24, -151.76 and –152.36 ppm with the coupling constant [1J (29Si-14N=33.04 Hz)]. Apparently, the chemical shifts are identical to those of pure silatrane. |
| d) Reaction of 1-Isothiocyanatosilatrane possessing six membered ring with copper(II) chloride |
| The reaction of equimolar quantities of 1-Isothiocyanato six membered silatrane and copper(II) chloride in dichloromethane medium at room temperature (25C) results in the formation of brown black solid. The compound isolated above is hygroscopic in nature and insoluble in the most of the solvents except dimethyl sulfoxide. Elemental analysis of this product corresponds to the composition CuCl2.SCNSi[(OC6H2Me2)CH2]3N. The reaction may be represented as follows (Scheme 4): |
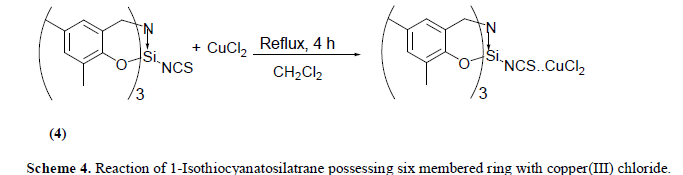 |
 |
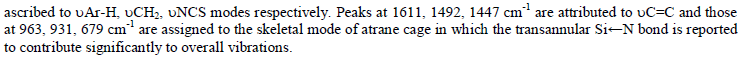 |
| NMR Spectra: The poor solubility of the adduct 4 in non-coordinating solvents has precluded detailed NMR studies of the adduct. However, the multinuclei (1H, 13C and 29Si) NMR spectra in DMSO-d6 solvent have been recorded in order to gather evidences in support of retention of silatrane moiety in the adduct. The results are described below. 1H NMR spectrum of the adduct 4 in DMSO-d6 reveals two singlets of equal intensity at 2.19 and 2.22 ppm due to aromatic methyl protons and a singlet 4.24 ppm due to NCH2, two singlets of equal intensity at 6.91and 6.93 ppm due to aromatic protons respectively. |
| 13C NMR spectrum of the adduct in DMSO-d6 reveals two singlets at 16.10 and 19.87 ppm due to aromatic methyl carbons, singlet at 55.11 ppm due to NCH2. The carbon atom of the isothiocyanato group appears as a triplet at 128.31, 128.52 & 128.67 ppm due to 13C-14N coupling [1J (13C-14N=13.50Hz)] and singlets at 116.07, 124.33, 129.01, 129.04 and 150.96 ppm appear due to aromatic carbons. |
| Similarly, the 29Si NMR spectrum of the complex reveals a triplet of equal intensity at–149.31, -149.86 and –150.44 ppm with the coupling constant [1J (29Si-14N=33.33 Hz)]. Apparently, the chemical shifts are identical to those of pure silatrane. |
| Table 1 summarizes the results obtained from the present study along with the possible mode of coordination of the title silatrane with Lewis acids. |
 |
V. CONCLUSIONS |
| The reactivity of 1-Isothiocyanato six membered towards Lewis acids (iron(III) chloride, nickel(II) chloride, cobalt(II) chloride, copper(II) chloride) has been studied successfully. The spectroscopic data has helped to establish possible site of coordination of 1-Isothiocyanato silatrane with various Lewis acids |
References |
|