ISSN ONLINE(2319-8753)PRINT(2347-6710)
ISSN ONLINE(2319-8753)PRINT(2347-6710)
S.A Thakur1, S.N.Nemade2, Sharanappa A.3
|
| Related article at Pubmed, Scholar Google |
Visit for more related articles at International Journal of Innovative Research in Science, Engineering and Technology
Biochemical conversion of agro-industrial wastes is an effective way of value addition. During the extraction of oil from soybean seed, due to supply of excessive heat or being retained for more time in desolventizer, the meal quality is reduced due to overheating and hence rejected for animal feed. The solid state fermentation using Aspergillus oryzae (NCIM No.649) to recover protease shows excellent potential of the overheated soybean meal (waste) as source. During the study the effect of varying particle size, pH, moisture content, incubation time and Protease activity has been measured.
Keywords |
| Solid state fermentation, Overheated soybean meal, Protease |
INTRODUCTION |
| The utilization of the agro industrial wastes, provides alternative substrates and, helps in solving pollution problems, which otherwise may cause problem in their disposal [1] The potential of solid state fermentation in converging agroindustrial residues into value added products is immense. Enzymes are the solution to replace traditional means of meeting industry needs. The cost effective, environment friendly enzymes helps towards more sustainable industrial development [2]. Agro-industrial substrates are considered best for enzyme production in solid state fermentation (SSF). The cost of enzyme production by submerged fermentation is higher compared to SSF [3]. The SSF process has the potential to significantly reduce the enzyme production costs because of lower energy requirements, increased productivity, smaller effluent volumes and simpler fermentation equipment. Agro-industrial residues are generally considered best substrates for the SSF processes and enzyme production by SSF is not an exception to that [4]. SSF uses agro-industrial waste as support and/or carbon source for production of various value added products, such as single cell protein, industrial enzymes, secondary metabolites, and fine chemicals [5]. |
| Oil-seed cakes are rich in protein and are recognized as being good food supplements, and some have been used for feed applications in poultry, fish and pig production. They also add value to various biotechnological processes such as the production of enzymes, antibiotics and mushrooms by fermentation [6]. Most soybean meal is produced by the solvent extraction process whereby the soybeans are cracked, heated and flaked before the oil is extracted with the solvent hexane. Once the oil has been removed, the flakes are toasted and ground into meal. During this production process, temperature is critical in order to deactivate the antinutritional factors naturally present in raw soybeans [7]. However, to maintain optimal nutritional value, the meal must not be subjected to excessive heat overheating of soybean meal reduces its nutritional value for poultry [8,9,10]. It has been shown that overcooking of soybean meal decreases digestibility of amino acids [11,12]. Excessive heat or heating time reduces the availability of amino acids due to the Maillard reaction [13]: and tends to destroy certain amino acids [14], the decreased amino acid digestibility and reduced growth responses appear to be related to the Maillard reaction with cross-linking involved to a lesser extent [12]. In addition to chemical composition, color differences are apparent with soybean meal subjected to overprocessing indicating a browning during the latter stage of Maillard reaction [15]. The overheated soybean meal due to browning and lower protein content has been rejected and hence the effective waste management is essential. |
| Protease is one of the most important commercial enzymes, and is used in food processing, detergents, diary industry and leather making [16]. Alkaline proteases (or Subtilisins, E.C. 3.4.21.14) are a physiologically and commercially important group of enzymes, play a specific catalytic role in the hydrolysis of proteins [17]. Proteases may be obtained from plant, animal or microbial sources but microbial sources are the most preferred ones due to multiple advantages including their broad biochemical diversity and bioengineering potentiality [18]. The fungal proteases are active over a wide pH range (4-11) and exhibit broad substrate specificity [19]. |
MICROORGANISM AND ENOCULUM |
| Aspergillus oryzae is widely used in Asiatic food fermentations; therefore, its proteases can be used in food processing and other applications [20]. Aspergillus oryzae has the ability to produce enzymes such as hemicelluloses, hydrolases, pectinases, lipases, and tannases and soybean hull as the substrate has shown presence of five kinds of cellullolytic enzyme activities [21]. Recent research results have shown that that fermentation of soybean hulls and meal with two microorganisms - Aspergillus oryzae and Bacillus spp. - degrade soybean hull fiber, increase soybean hull protein content, degrade oligosaccharides in soybean meal, and degrade soybean meal protein peptide sizes [22]. Aspergillus oryzae (NCIM No.649) was maintained on potato dextrose agar (PDA) slants of pH 5.6 at 4OC. After tray drying and grinding the substrate was prepared to suitable mesh size by screening. 10 gm each of above grinded substrate taken in different 250 ml conical flask or petriplates was moistened with 15 ml phosphate buffer (7.2 pH) each. Moistened substrate was taken in to autoclave and sterilized for 15 minute at 121OC for proper cooking of the substrate and to increase its amenability for microorganisms. Aspergillus oryzae (NCIM No.649) spores were transferred aseptically and separately to potato dextrose broath, in a 100 ml conical flask containing 50 ml of sterilized inoculum medium (sterilized at 121OC for 15 minutes) in laminar air flow. The flask was then kept in incubator at 37O C for 48 hrs. The homogenous spores suspension (106 –107 spores / ml) was used as inoculums [23,24]. Autoclave was used for sterilizing glass wares and media to avoid contamination of undesired microorganism. |
SOLID STATE FERMENTATION |
| For Solid State Fermentation the substrate overheated soybean meal and was collected from Maharashtra Oil Extraction Pvt. Ltd., Dhule (Maharashtra, India). The substrate was pre-characterized as Oil Content, 0.8%, Moisture Content 7.8%, Protein Content 42%, Fiber Content 9.4%, and Ash Content 2.4%. After sterilizing the substrate, the substrate was cooled to room temperature. The substrate of 10 gm in petriplate and 15 gm in conical flask of 250 ml were added with the inoculum of 30 % (W/V) in the laminar air flow with the help of sterilized pipette. |
| Aspergillus oryzae (NCIM No.649) were inoculated on overheated soybean meal (waste). After inoculation, the flask and petriplates were then incubated at 37OC for 2 days. The SSF media flasks and petriplates were gently shaken after every 12 hrs for uniform mixing of the substrate and micro organism. |
| The fermented overheated soybean meal samples were extracted with 1:10 (W/ V) of 0.1 M sodium phosphate buffer of pH 6.9 was added to each conical flask. The fermented substrate was first taken in 250 ml conical flask in laminar air flow and then buffer was added. The flask was shaken at 150 rpm for 60 minute and material was filtered through muslin cloth or was filtered through whatman filter paper. Filtrate collected was centrifuged at 1000 rpm for 10 minutes at room temperature. Supernatant was carefully collected and used as crude enzyme extract for determining protease activity [25]. |
| Protease activity was determined by a modified Anson’s method. Protein was estimated by Lowry’s method. The blue color developed by the reduction of phosphomolybdic phosphotungstic components in the folin-ciocalteau reagent by amino acids tyrosine and tryptophan present in the protein plus the color developed by the biuret reaction of the protein with the alkaline cupric tartarate were measured in the Lowry’s method. |
RESULTS AND DISCUSSION |
| The effect of varying particle size, pH, moisture content, incubation time and protease activity was investigated during the study. |
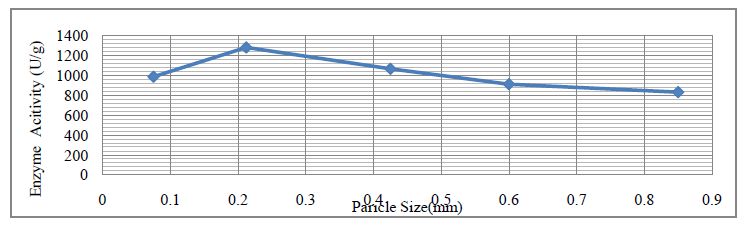
| Effect of particle size on protease activity was studied by taking overheated soybean meal (waste) samples as substrate. After grinding the substrate the substrate of different particle size from 0.075 mm to 0.850 mm was taken to study the effect of particle size on enzyme activity. Sieve shaker was used to separate the substrate particles of different size. Substrate of each particle size was taken in conical flask (250 ml) and SSF was carried out for 48 hours at 37OC. The crude enzyme was extracted; a reading of each particle size was recorded for enzyme activity. The graph of mean reading of enzyme activity (shown in percentage) against particle size is shown in figure 1. It has been observed that particle size 0.212mm provide better respiration/aeration and digestion efficiency due to small surface area. In contrast, a large substrate particle or lumps may result in interfere with microbial respiration/aeration and poor digestion of raw material and therefore result in poor growth and enzyme production. For protease activity, decrease in particle size from 0.850 mm to 0.212 mm shows increase in enzyme activity, but further decrease in particle to 0.075 mm decreases enzyme activity. Optimal activity of 1283 U/g was seen in 0.212 mm particle size. |
| Figure 1: Effect of Particle Size on Protease Activity |
 |
| To investigate the effect of pH, moisture content, incubation period and Protease activity particle size 0.212 mm was used |
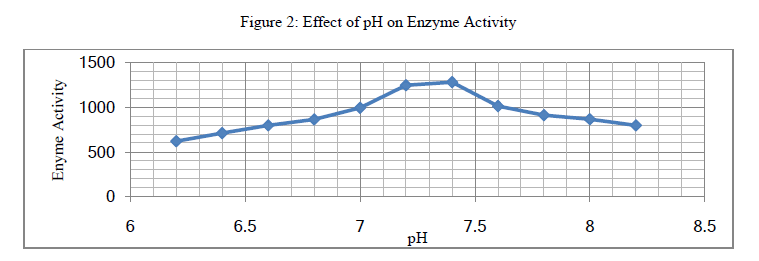
| 1. Effect of pH on Enzyme Activity: SSF was performed to study the effect of incubation pH on enzyme activity. A moistened overheated Soybean Meal (waste) had an initial pH of about 6.2. Phosphate buffer (pH 7.2) was used to adjust the initial pH to values greater than 6.2 in separate fermentations. For protease activity, increase in pH from 6 to 7.4 increases enzyme activity, further increase in pH decreases activity. Optimal activity of 1281 U/g was observed at pH 7.4 in fig. 2. |
 |
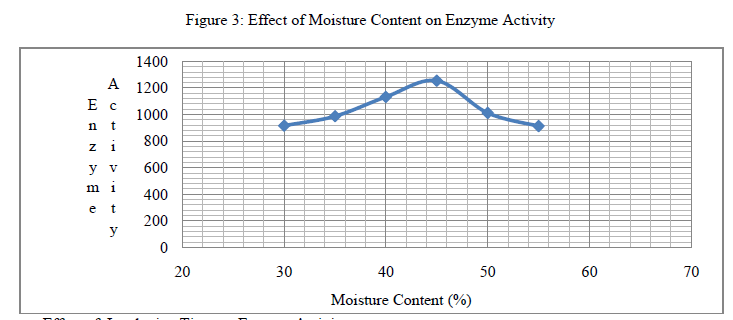
| 2. Effect of Moisture Content on Enzyme Activity: Initial moisture contents of the substrate are known to critically influence mold growth and enzyme production. The impact on physicochemical properties of the substrate affects enzyme production. Presence of water in the substrate makes the nutrients more easily accessible for mold growth. Too much water adversely affects oxygen diffusion in the substrate. In this work, initial moisture contents of the substrate were adjusted to 30, 35, 40, 45, 50, and 55 % in separate experiments before inoculation with spores. According to results the optimum initial moisture level was about 45% and the protease activity was observed as 1254 U/g. |
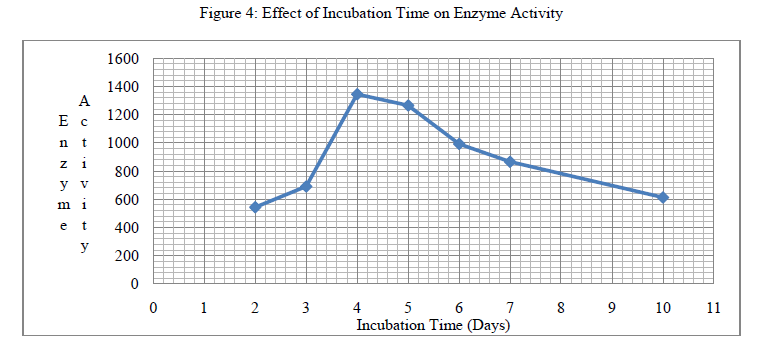
| 3. Effect of Incubation Time on Enzyme Activity: SSF was performed by varying incubation period from 2 to 7 days at 37OC. Increase in incubation period from 2 days to 4 days increases protease activity, whereas increase in incubation period from 5 days and above shown decrease in protease activity. The optimal activity of 1345.14 U/g was observed at 96 hours. |
 |
| 3. Effect of Incubation Time on Enzyme Activity: SSF was performed by varying incubation period from 2 to 7 days at 37OC. Increase in incubation period from 2 days to 4 days increases protease activity, whereas increase in incubation period from 5 days and above shown decrease in protease activity. The optimal activity of 1345.14 U/g was observed at 96 hours. |
 |
| 4. Determination of Protease Activity: Enzyme protein concentration was estimated by Lowry’s Method. The absorbance optical density was measured using UV spectrophotometer for different particle size and the Protease activity was calculated. |
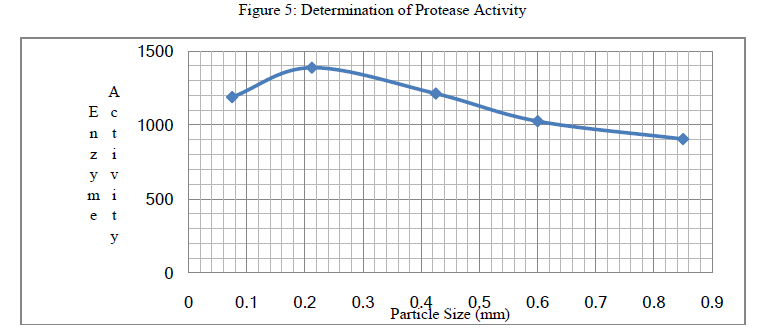 |
| The Protease activity was recorded has shown a decrease in the enzyme activity with increase in the particle size of the substrate. |
CONCLUSION |
| Overheated Soybean Meal (waste) has shown a good potential for recovery of protease as a value added product, providing effective agro-industrial waste remediation. The maximum activity of protease produced by Aspergillus oryzae (NCIM No.649) on Soybean Meal (Overheated) for particle size (1283 U/g for 0.212mm), incubation pH (1281 U/g for pH 7.4), incubation period (1345.14 U/g on 4th day), moisture content (1254 U/g for 45%) and protease activity (1387.6 U/g) was recorded. |
References |
|